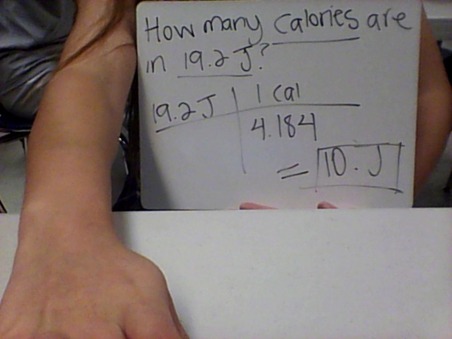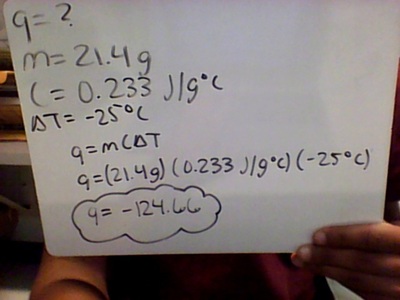Units of Heat:
Temperature vs. Heat - Temperature can be measured with thermometers but heat cannot be measured. We can only measure the changes in heat.
Heat Capacity - the heat capacity is dependent on the mass and the chemical composition of a substance
- 1 Calorie= 1000 calories= 1 Kilo calorie
- 1 calorie= 4.184 joules. 1 Kilo joule= 1000 joules
- q= heat
- m= mass
- C= specific heat
- ΔT- change in temperature (final temperature-Initial temperature)
Temperature vs. Heat - Temperature can be measured with thermometers but heat cannot be measured. We can only measure the changes in heat.
Heat Capacity - the heat capacity is dependent on the mass and the chemical composition of a substance
 Formula set up for heat calculation equations
Formula set up for heat calculation equations
Heat and Energy: https://www.youtube.com/watch?v=D30gwbupVlc&index=1&list=PLB2cIJBZdJjTEhjT4pLQK6e0kygnXoY0F
Examples of equations:
Sources Used:
Chemistry 10.1 Heat and Energy. YouTube. YouTube, 8 Mar. 2012. Web. 28 May 2014.Cool, Stacy. "Lesson 8.A-Heat." Cool's Chemistry. Web. 28 May 2014. <http://www.coolchemteacher.org/lesson-8a---heat.html>.
Cool, Stacy. "Lesson 8.B- Specific Heat." Cool's Chemistry. Web. 28 May 2014. <http://www.coolchemteacher.org/lesson-8b---specific-heat.html>.
Chemistry 10.1 Heat and Energy. YouTube. YouTube, 8 Mar. 2012. Web. 28 May 2014.Cool, Stacy. "Lesson 8.A-Heat." Cool's Chemistry. Web. 28 May 2014. <http://www.coolchemteacher.org/lesson-8a---heat.html>.
Cool, Stacy. "Lesson 8.B- Specific Heat." Cool's Chemistry. Web. 28 May 2014. <http://www.coolchemteacher.org/lesson-8b---specific-heat.html>.